Opinion: The podcast telling the stories behind Ambien, Ozempic, EpiPens, and other game-changing drugs

Saturday, April 25, 2026 – Saturday, April 25 - “We're basically not using our body as directed,” says "Drug Story" host Thomas Goetz.

Saturday, April 25, 2026 – Saturday, April 25 - “We're basically not using our body as directed,” says "Drug Story" host Thomas Goetz.
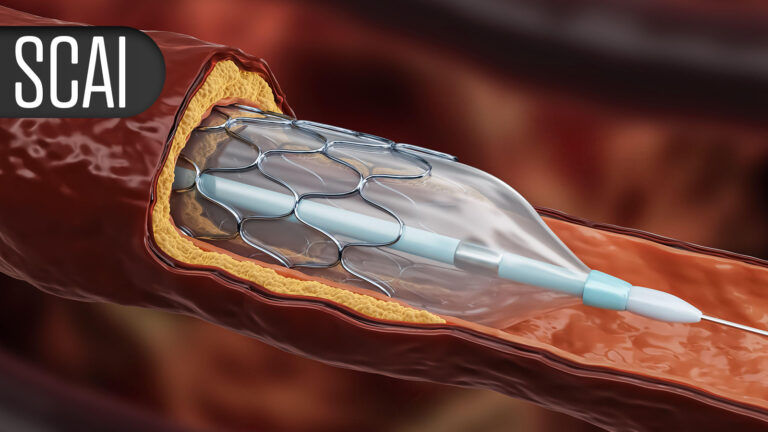
Friday, April 24, 2026 – Saturday, April 25 - (MedPage Today) -- Cardiovascular protection with GLP-1 receptor agonists may benefit patients undergoing certain transcatheter procedures, according to two observational studies.
When used as an adjunct to transcatheter aortic valve replacement...

Friday, April 24, 2026 – Saturday, April 25 - The new generation of weight loss medicines does an unprecedented job of keeping off the pounds — if people stay on the drugs. Yet numerous studies suggest most of those who start taking GLP-1 drugs quit ...

Friday, April 24, 2026 – Saturday, April 25 - Teddi Mellencamp says her doctor denied her request for GLP-1s after steroid weight gain during her stage 4 melanoma treatment in a podcast discussion.
Friday, April 24, 2026 – Saturday, April 25 - GLP-1 drugs containing tirzepatide significantly reduce cardiovascular risks in people with serious heart conditions. Indranil MUKHERJEE / AFP via Getty Images
GLP-1 weight loss medications containing tirzepatide (Mounjaro, Zepbound) can reduce heart-related health risks for people with serious heart conditions, according to new research.
Thursday, April 23, 2026 – Friday, April 24 - (MedPage Today) -- Welcome to Culture Clinic, MedPage Today's collaboration with Northwell Health to offer a healthcare professional's take on the latest viral medical topics.
Many patients taking glucagon-like peptide-1 (GLP-1) receptor agonists...
Thursday, April 23, 2026 – Friday, April 24 - An outpatient procedure may offer a way for the estimated 70% of people who discontinue popular weight-loss drugs to avoid regaining the pounds they shed, according to a study presented at Digestive Disease Week (DDW) 2026.

Thursday, April 23, 2026 – Friday. April 24 - Bagsværd, Denmark, – Novo Nordisk today announced positive topline results from PIONEER TEENS, a phase 3a trial evaluating oral semaglutide for type 2 diabetes in children and adolescents aged 10–17 years with type 2...

Thursday, April 23, 2026 – Thursday, April 23 - Roche and Zealand Pharma announced last month that their amylin analog petrelintide elicited a 9% placebo-controlled weight reduction at 42 weeks—falling far below analyst and investor expectations.

Thursday, April 23, 2026 – Thursday, April 23 - A new minimally invasive procedure may help people keep weight off after stopping popular drugs like Ozempic and semaglutide—something most patients struggle with. In a clinical trial, those who underwent a technique called duodenal mucosal resurfacing regained far less weight compared to others after discontinuing the medication. The procedure works by renewing the lining of the upper small intestine, potentially “resetting” metabolism and preserving the benefits of weight loss.