Oncobesity News Posts

What is ‘food noise’? UK surgeon explains why people with obesity are more reactive to food; shares 5 ways to curb it
Sunday, February 1, 2026 – “Food noise” is a real thing where some brains experience heightened reward sensitivity associated with food. Dr Rajan explains how GLP-1 treatment can help.

Summary of JP Morgan Conference in Obesity and Metabolic Diseases
Friday, January 16, 2026 – Here’s a summary of the major announcements and trends from the 44th Annual JP Morgan Healthcare Conference in San Francisco (January 12–15, 2026) specifically in

Day 1 @ JP Morgan in Obesity Drugs and Therapies
Friday, January 16, 2026 – JP Morgan 2026: Obesity & GLP-1 Therapies Take Center StageWhat the Latest Deals and Data Mean for the Future of Metabolic MedicineThe J.P. Morgan Healthcare

Day 2 @ JP Morgan in Obesity Drugs and Therapies
Friday, January 16, 2026 – Here are the **highlights from Day 2 of the JP Morgan Healthcare Conference (San Francisco) specifically related to obesity, weight-gain therapies, and related drug developments

Cardiologist explains benefits of GLP-1 drugs go beyond just weight loss: ‘Future of how we treat heart, liver…’
Thursday, January 15, 2026 – Everyone’s talking about weight loss drugs like Ozempic, Wegovy and Mounjaro and their implications on weight loss – but their benefits go way beyond that.

JP Morgan Healthcare Conference – Day 3 Highlights (Obesity & Metabolic Therapies)
Wednesday, January 14, 2026 – JP Morgan Healthcare Conference – Day 3 Highlights (Obesity & Metabolic Therapies)1. Shift From “Weight Loss” to Chronic Metabolic DiseaseDay 3 discussions made it clear

Cardiologist with 20 years of experience shares the truth behind GLP-1 drugs and weight loss: ‘Disaster always follows…’
Wednesday, January 14, 2026 – Dr Bhojraj explains that GLP-1 isn’t a medication but a natural hormone, and while it can help, they have side effects and aren’t meant for

Does Ozempic need to be taken forever once started? UK surgeon explains the effects of stopping GLP-1 drugs
Monday, January 12, 2026 – According to Dr Rajan, GLP-1 drugs such as Ozempic is not designed for long-term weight loss, which requires a comprehensive approach.

Is ozempic forever? UK surgeon explains how GLP-1 drugs work, and whether you need to take them indefinitely
Friday, December 5, 2025 – If you are considering GLP-1 drugs for weight loss, it might be a more serious commitment than you expected. Dr Rajan explains how they work.

Struggling with chronic stress? Wellness coach shares 7 nutrients to lower cortisol and boost GLP-1 naturally
Friday, December 5, 2025 – Chronic stress not only makes you feel overwhelmed but can also deplete key nutrients and mess with hormones. Rachel shares 7 nutrients that can help.

Urgent warning issued for Ozempic after GLP-1 drugs linked to suicide
Monday, December 1, 2025 – New precautions added for GLP-1 drugs as reports of mood changes and contraceptive concerns emerge in Australia.
Cardiovascular benefits of GLP-1 drugs independent of weight loss
Saturday, November 22, 2025 – Revising our earlier interpretation of SELECT trial data in light of new analyses The post Cardiovascular benefits of GLP-1 drugs independent of weight loss appeared
Itching at the GLP-1 site of injection
Tuesday, November 11, 2025 – Some people will get itching at the site of injection of the GLP-1 drugs. This is not common but is irritating. There are some things you can do to lessen the severity of the reaction.

Trump Cuts Prices on Ozempic and Wegovy in Deal With GLP-1 Weight Loss Drug Makers
Thursday, November 6, 2025 – President Trump’s plan drops the price of Ozempic and Wegovy for Americans, with plans for Medicare to cover the popular GLP-1 weight loss drugs.
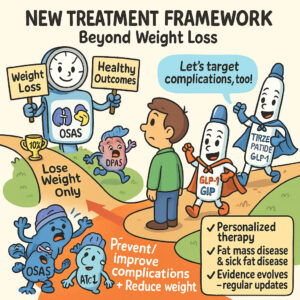
EASO Framework for Pharmacological Obesity Treatment – Sick Fat vs. Fat Mass
Friday, October 31, 2025 – A framework published by the European Association for the Study of Obesity (EASO) that addresses the pharmacological treatment of obesity and its complications. This framework proposes a new

Surgery beats Ozempic for long-term health, Cleveland Clinic finds
Saturday, October 18, 2025 – Weight-loss surgery dramatically outperformed GLP-1 medications in improving longevity and reducing heart, kidney, and eye complications for people with obesity and diabetes. Over 10 years, patients lost far more weight and required fewer medications. Experts say surgery continues to offer survival advantages even in the age of potent obesity drugs.
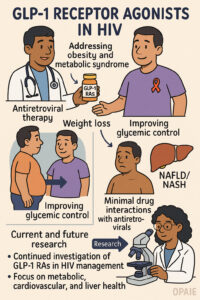
💪 The HIV and Weight Loss Drugs Connection – GLP-1 Agonists in HIV Management
Sunday, August 3, 2025 – The provided source explores the increasing use of GLP-1 receptor agonists like semaglutide and liraglutide in individuals with HIV. It highlights that these medications are beneficial for addressing obesity

💲 Medicare’s Weight Loss Drug Experiment: Costs & Coverage
Sunday, August 3, 2025 – A Washington Post article talks about the Trump administration experiment to expand Medicare and Medicaid coverage for GLP-1 weight loss drugs like Ozempic, potentially benefiting millions of Americans
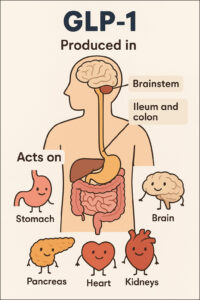
Where is GLP-1 is produced and where in the body does it act?
Friday, August 1, 2025 – The provided text describes glucagon-like peptide-1 (GLP-1), detailing its production and the wide array of organs and systems it affects. Primarily, GLP-1 is generated in the intestinal

⚖️ GLP-1s: Integrating Diet and Activity for Optimal Outcomes
Thursday, July 31, 2025 – This JAMA article provides comprehensive guidance for clinicians on integrating diet and physical activity when prescribing GLP-1 receptor agonists for weight management. It outlines strategies for monitoring and

⚖️ Navigating GLP-1 Weight Loss: Diet, Exercise, and Well-being
Saturday, July 26, 2025 – This patient-focused guide from JAMA Internal Medicine provides essential information for individuals utilizing GLP-1 medications for weight loss. It outlines the medications’ functions and offers practical dietary advice through a “MEAL” plan, emphasizing muscle

🧪 Unregulated Additives in Compounded GLP-1 Medications
Wednesday, July 23, 2025 – The practice of compounding pharmacies adding various supplements to GLP-1 receptor agonist formulations like semaglutide or tirzepatide is very common in the United States. These additions, which

⚠️ FDA Failures and GLP-1 Compounding Risks
Tuesday, July 22, 2025 – “FDA Regulatory Failures in Enforcing Limits on GLP-1 Compounding Puts Patients at Risk,” critically examines the Food and Drug Administration’s (FDA) oversight of compounded GLP-1 medications.

🤢 Adiposity and Colorectal Cancer Risk: A Systematic Review
Sunday, July 20, 2025 – A systematic review investigating the relationship between adiposity measures and the risk of developing colorectal cancer (CRC), building upon existing research that often relies solely on Body Mass

💪 GLP-1s: Diabetes, Weight Loss, and Cancer Reduction
Friday, July 18, 2025 – This source explores the impact of semaglutide, a weight loss medication, on muscle mass, glucose regulation, and bone health in adults with obesity. It highlights

💲 Tirzepatide: A Cost-Effective Path to Obesity Management
Thursday, July 17, 2025 – This academic article presents a cost-effectiveness analysis of tirzepatide, a medication for managing overweight and obesity, comparing it against lifestyle modification (LSM) alone. Researchers used a simulation model to project lifetime health

🏃 Physical Activity Trajectories and Mortality: A Meta-Analysis
Wednesday, July 16, 2025 – A systematic review and meta-analysis of 85 prospective studies, examining the long-term impact of physical activity (PA) patterns throughout adulthood on mortality. The authors aimed to understand how consistent, increasing,

Hormone Therapy Enhances Tirzepatide Weight Loss in Menopause
Tuesday, July 15, 2025 – The source discusses research indicating that postmenopausal women on hormone therapy (HT) experience significantly greater weight loss when treated with tirzepatide, an obesity medication, compared

⚖️ The History of Obesity
Sunday, July 13, 2025 – The provided text offers a comprehensive overview of how obesity has been understood and depicted across different historical periods, from ancient civilizations to the present day. It traces

👨⚕️ AI-Driven Virtual Human for GP Obesity Training
Saturday, July 12, 2025 – This document presents a pilot feasibility study published in “Obesity Science & Practice” that investigates the effectiveness of an AI-driven Virtual Human (VH) intervention designed to enhance General Practitioner (GP)

👵 Ozempic Face: GLP-1 and Aesthetic Surgery
Friday, July 11, 2025 – This scholarly article from Aesthetic Surgery Journal Open Forum discusses the phenomenon of “Ozempic Face,” a term used to describe the accelerated facial aging and volume loss experienced by some patients

✨ GLP-1s and Post-Weight Loss Plastic Surgery Trends
Thursday, July 10, 2025 – There is growing evidence—especially in the plastic surgery community—that the recent surge in GLP‑1 medications (like Ozempic, Wegovy, Mounjaro) for weight loss is fueling a

💉 Counterfeit Ozempic: FDA Warns of Unsafe Drug Supply (4/14/2025)
Monday, July 7, 2025 – The provided text, originating from the Food and Drug Administration (FDA), serves as a crucial public health warning regarding counterfeit Ozempic (semaglutide) injections found within the U.S. drug

Obesity Treatment Outcomes with Semaglutide or Tirzepatide
Saturday, July 5, 2025 – This original research article examines the real-world effectiveness of semaglutide and tirzepatide for obesity treatment, focusing on how medication discontinuation impacts weight loss and glycemic control. The retrospective cohort study analyzed electronic health records from

Unpacking the GLP-1 Gold Rush
Monday, June 30, 2025 – We ask experts for their views on the GLP-1 market in this exclusive eBook. GLP-1 receptor agonists have rapidly evolved from niche diabetes drugs to
Your Weight Matters Region
Monday, June 30, 2025 – Your Weight Matters Regional is traveling the country, bringing expert-led education and practical guidance to cities near you. These FREE half-day events are designed to
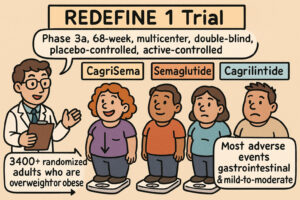
CagriSema: Weight Loss in Overweight or Obese Adults
Tuesday, June 24, 2025 – This document details a Phase 3a, 68-week, multicenter, double-blind, placebo-controlled, and active-controlled clinical trial (REDEFINE 1) investigating the efficacy and safety of cagrilintide–semaglutide (CagriSema) for weight loss in adults

GLP-1 Agonists: Uses, Mechanism, and Side Effects
Tuesday, June 24, 2025 – https://my.clevelandclinic.org/health/treatments/13901-glp-1-agonists The provided text from the Cleveland Clinic offers a comprehensive overview of GLP-1 agonists, a class of medications primarily used to manage Type 2 diabetes and obesity. It explains how these

Orforglipron for Early Type 2 Diabetes Management
Sunday, June 22, 2025 – This scientific paper presents the ACHIEVE-1 trial, a phase 3 study investigating orforglipron, an oral, nonpeptide GLP-1 receptor agonist, for treating type 2 diabetes. The research evaluates its efficacy

ADA (American Diabetes Association) June 20-23 Obesity Medicines – Other Posters and Talks
Thursday, June 19, 2025 – 🧪 NA‑931 (Bioglutide™) – Biomed Industries, Inc.Presentation (Oral):Title: “Phase 2 Clinical Trials of NA‑931, an Oral Novel Quadruple IGF‑1, GLP‑1, GIP, and Glucagon Receptor Agonist,
